(02.03 Mc) How Does The Location Of Dna In Animal Cells Compare To Dna's Location In Plant Cells?

| Cell biology | |
|---|---|
| Animal cell diagram | |
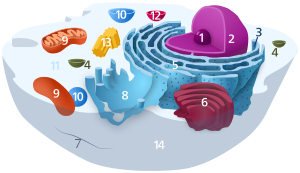 Components of a typical creature cell:
|
In cell biology, the nucleus (pl. nuclei ; from Latin nucleus or nuculeus , pregnant kernel or seed) is a membrane-jump organelle found in eukaryotic cells. Eukaryotes usually take a single nucleus, merely a few cell types, such as mammalian red blood cells, have no nuclei, and a few others including osteoclasts have many. The main structures making up the nucleus are the nuclear envelope, a double membrane that encloses the entire organelle and isolates its contents from the cellular cytoplasm; and the nuclear matrix (which includes the nuclear lamina), a network within the nucleus that adds mechanical support, much like the cytoskeleton supports the prison cell as a whole.
The cell nucleus contains all of the prison cell'southward genome, except for the small amount of mitochondrial DNA and, in constitute cells, plastid Dna. Nuclear Dna is organized as multiple long linear molecules in a complex with a large diverseness of proteins, such as histones, to class chromosomes. The genes within these chromosomes are structured in such a way to promote cell part. The nucleus maintains the integrity of genes and controls the activities of the cell by regulating gene expression—the nucleus is, therefore, the control center of the cell.
Because the nuclear envelope is impermeable to large molecules, nuclear pores are required to regulate nuclear send of molecules across the envelope. The pores cross both nuclear membranes, providing a channel through which larger molecules must be actively transported by carrier proteins while assuasive gratuitous movement of small-scale molecules and ions. Movement of large molecules such equally proteins and RNA through the pores is required for both gene expression and the maintenance of chromosomes.
Although the interior of the nucleus does not contain whatever membrane-leap subcompartments, its contents are not uniform, and a number of nuclear bodies exist, made up of unique proteins, RNA molecules, and item parts of the chromosomes. The best-known of these is the nucleolus, which is mainly involved in the assembly of ribosomes. After being produced in the nucleolus, ribosomes are exported to the cytoplasm where they translate messenger RNA.
Structures

The nucleus contains nearly all of the prison cell's DNA, surrounded by a network of gristly intermediate filaments and enveloped in a double membrane called the "nuclear envelope". The nuclear envelope separates the fluid inside the nucleus, called the nucleoplasm, from the rest of the cell. The size of the nucleus depends on the size of the cell it is independent in, with a nucleus typically occupying about 8% of the full cell volume.[ane] The nucleus is the largest organelle in brute cells.[2] : 12 In mammalian cells, the average bore of the nucleus is approximately 6 micrometres (µm).[3]
Nuclear envelope and pores
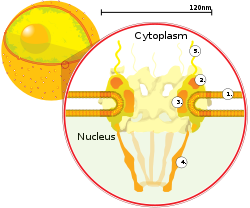
A cantankerous department of a nuclear pore on the surface of the nuclear envelope (1). Other diagram labels show (2) the outer ring, (3) spokes, (4) basket, and (5) filaments.
The nuclear envelope consists of two membranes, an inner and an outer nuclear membrane.[4] : 649 Together, these membranes serve to split the prison cell's genetic material from the residuum of the prison cell contents, and allow the nucleus to maintain an surround distinct from the rest of the cell. Despite their shut apposition around much of the nucleus, the 2 membranes differ substantially in shape and contents. The inner membrane surrounds the nuclear content, providing its defining edge.[2] : xiv Embedded within the inner membrane, diverse proteins bind the intermediate filaments that requite the nucleus its structure.[4] : 649 The outer membrane encloses the inner membrane, and is continuous with the adjacent endoplasmic reticulum membrane.[4] : 649 As part of the endoplasmic reticulum membrane, the outer nuclear membrane is studded with ribosomes that are actively translating proteins across membrane.[4] : 649 The space betwixt the two membranes, called the "perinuclear space", is continuous with the endoplasmic reticulum lumen.[4] : 649
Nuclear pores, which provide aqueous channels through the envelope, are composed of multiple proteins, collectively referred to as nucleoporins. The pores are about 60–80 meg daltons in molecular weight and consist of around fifty (in yeast) to several hundred proteins (in vertebrates).[2] : 622–4 The pores are 100 nm in total diameter; all the same, the gap through which molecules freely diffuse is only about 9 nm wide, due to the presence of regulatory systems inside the center of the pore. This size selectively allows the passage of minor h2o-soluble molecules while preventing larger molecules, such as nucleic acids and larger proteins, from inappropriately entering or exiting the nucleus. These large molecules must exist actively transported into the nucleus instead. The nucleus of a typical mammalian prison cell volition have about 3000 to 4000 pores throughout its envelope,[five] each of which contains an eightfold-symmetric band-shaped structure at a position where the inner and outer membranes fuse.[vi] Attached to the band is a structure called the nuclear basket that extends into the nucleoplasm, and a series of filamentous extensions that reach into the cytoplasm. Both structures serve to mediate bounden to nuclear transport proteins.[seven] : 509–10
Most proteins, ribosomal subunits, and some RNAs are transported through the pore complexes in a process mediated by a family of transport factors known every bit karyopherins. Those karyopherins that mediate movement into the nucleus are also called importins, whereas those that mediate movement out of the nucleus are called exportins. Most karyopherins interact directly with their cargo, although some employ adaptor proteins.[viii] Steroid hormones such as cortisol and aldosterone, also as other small lipid-soluble molecules involved in intercellular signaling, can diffuse through the cell membrane and into the cytoplasm, where they bind nuclear receptor proteins that are trafficked into the nucleus. There they serve as transcription factors when bound to their ligand; in the absence of a ligand, many such receptors function every bit histone deacetylases that repress gene expression.[7] : 488
Nuclear lamina
In animate being cells, ii networks of intermediate filaments provide the nucleus with mechanical support: The nuclear lamina forms an organized meshwork on the internal face of the envelope, while less organized support is provided on the cytosolic face of the envelope. Both systems provide structural support for the nuclear envelope and anchoring sites for chromosomes and nuclear pores.[nine]
The nuclear lamina is composed mostly of lamin proteins. Like all proteins, lamins are synthesized in the cytoplasm and later transported to the nucleus interior, where they are assembled before beingness incorporated into the existing network of nuclear lamina.[x] [eleven] Lamins found on the cytosolic face of the membrane, such as emerin and nesprin, bind to the cytoskeleton to provide structural support. Lamins are also found within the nucleoplasm where they form another regular structure, known as the nucleoplasmic veil,[12] [13] that is visible using fluorescence microscopy. The actual function of the veil is not clear, although information technology is excluded from the nucleolus and is nowadays during interphase.[14] Lamin structures that brand up the veil, such as LEM3, demark chromatin and disrupting their structure inhibits transcription of protein-coding genes.[xv]
Like the components of other intermediate filaments, the lamin monomer contains an alpha-helical domain used by two monomers to coil effectually each other, forming a dimer structure called a coiled coil. Two of these dimer structures so join adjacent, in an antiparallel arrangement, to form a tetramer called a protofilament. Eight of these protofilaments grade a lateral arrangement that is twisted to form a ropelike filament. These filaments tin can be assembled or disassembled in a dynamic manner, significant that changes in the length of the filament depend on the competing rates of filament addition and removal.[9]
Mutations in lamin genes leading to defects in filament assembly cause a group of rare genetic disorders known as laminopathies. The most notable laminopathy is the family of diseases known every bit progeria, which causes the appearance of premature crumbling in its sufferers. The exact machinery by which the associated biochemical changes give rise to the anile phenotype is not well understood.[16]
Chromosomes

The cell nucleus contains the majority of the prison cell'southward genetic material in the grade of multiple linear DNA molecules organized into structures called chromosomes. Each human jail cell contains roughly 2 meters of Dna.[7] : 405 During most of the cell cycle these are organized in a Dna-protein complex known as chromatin, and during cell partition the chromatin tin can be seen to form the well-divers chromosomes familiar from a karyotype. A small fraction of the cell's genes are located instead in the mitochondria.[7] : 438
There are two types of chromatin. Euchromatin is the less compact DNA form, and contains genes that are frequently expressed past the jail cell.[17] The other type, heterochromatin, is the more meaty form, and contains DNA that is infrequently transcribed. This construction is further categorized into facultative heterochromatin, consisting of genes that are organized as heterochromatin merely in certain cell types or at certain stages of development, and constitutive heterochromatin that consists of chromosome structural components such as telomeres and centromeres.[eighteen] During interphase the chromatin organizes itself into detached individual patches,[xix] chosen chromosome territories.[20] Agile genes, which are more often than not establish in the euchromatic region of the chromosome, tend to be located towards the chromosome'south territory purlieus.[21]
Antibodies to sure types of chromatin organization, in particular, nucleosomes, have been associated with a number of autoimmune diseases, such every bit systemic lupus erythematosus.[22] These are known equally anti-nuclear antibodies (ANA) and have also been observed in concert with multiple sclerosis as part of full general immune system dysfunction.[23]
Nucleolus

The nucleolus is the largest of the detached densely stained, membraneless structures known equally nuclear bodies found in the nucleus. It forms around tandem repeats of rDNA, DNA coding for ribosomal RNA (rRNA). These regions are chosen nucleolar organizer regions (NOR). The master roles of the nucleolus are to synthesize rRNA and gather ribosomes. The structural cohesion of the nucleolus depends on its activity, as ribosomal assembly in the nucleolus results in the transient association of nucleolar components, facilitating further ribosomal assembly, and hence farther association. This model is supported by observations that inactivation of rDNA results in intermingling of nucleolar structures.[24]
In the kickoff stride of ribosome associates, a protein called RNA polymerase I transcribes rDNA, which forms a large pre-rRNA precursor. This is cleaved into ii large rRNA subunits – 5.8S, and 28S, and a small rRNA subunit 18S.[four] : 328 [25] The transcription, mail service-transcriptional processing, and associates of rRNA occurs in the nucleolus, aided by minor nucleolar RNA (snoRNA) molecules, some of which are derived from spliced introns from messenger RNAs encoding genes related to ribosomal function. The assembled ribosomal subunits are the largest structures passed through the nuclear pores.[7] : 526
When observed under the electron microscope, the nucleolus tin can be seen to consist of 3 distinguishable regions: the innermost fibrillar centers (FCs), surrounded by the dense fibrillar component (DFC) (that contains fibrillarin and nucleolin), which in turn is bordered by the granular component (GC) (that contains the protein nucleophosmin). Transcription of the rDNA occurs either in the FC or at the FC-DFC boundary, and, therefore, when rDNA transcription in the cell is increased, more than FCs are detected. Most of the cleavage and modification of rRNAs occurs in the DFC, while the latter steps involving protein associates onto the ribosomal subunits occur in the GC.[25]
Other nuclear bodies
| Structure name | Structure diameter | Ref. |
|---|---|---|
| Cajal bodies | 0.2–2.0 µm | [26] |
| Clastosomes | 0.ii-0.5 µm | [27] |
| PIKA | 5 µm | [28] |
| PML bodies | 0.2–1.0 µm | [29] |
| Paraspeckles | 0.5–1.0 µm | [30] |
| Speckles | 20–25 nm | [28] |
Also the nucleolus, the nucleus contains a number of other nuclear bodies. These include Cajal bodies, gemini of Cajal bodies, polymorphic interphase karyosomal association (PIKA), promyelocytic leukaemia (PML) bodies, paraspeckles, and splicing speckles. Although footling is known about a number of these domains, they are significant in that they show that the nucleoplasm is not a uniform mixture, just rather contains organized functional subdomains.[29]
Other subnuclear structures appear every bit role of abnormal affliction processes. For instance, the presence of pocket-sized intranuclear rods has been reported in some cases of nemaline myopathy. This condition typically results from mutations in actin, and the rods themselves consist of mutant actin equally well every bit other cytoskeletal proteins.[31]
Cajal bodies and gems
A nucleus typically contains between one and 10 compact structures called Cajal bodies or coiled bodies (CB), whose diameter measures between 0.2 µm and 2.0 µm depending on the cell type and species.[26] When seen under an electron microscope, they resemble balls of tangled thread[28] and are dense foci of distribution for the protein coilin.[32] CBs are involved in a number of different roles relating to RNA processing, specifically small nucleolar RNA (snoRNA) and small nuclear RNA (snRNA) maturation, and histone mRNA modification.[26]
Similar to Cajal bodies are Gemini of Cajal bodies, or gems, whose name is derived from the Gemini constellation in reference to their shut "twin" relationship with CBs. Gems are similar in size and shape to CBs, and in fact are about indistinguishable under the microscope.[32] Unlike CBs, gems do not contain modest nuclear ribonucleoproteins (snRNPs), but practice incorporate a protein called survival of motor neuron (SMN) whose function relates to snRNP biogenesis. Gems are believed to assistance CBs in snRNP biogenesis,[33] though it has also been suggested from microscopy testify that CBs and gems are unlike manifestations of the same construction.[32] Later ultrastructural studies have shown gems to exist twins of Cajal bodies with the difference beingness in the coilin component; Cajal bodies are SMN positive and coilin positive, and gems are SMN positive and coilin negative.[34]
PIKA and PTF domains
PIKA domains, or polymorphic interphase karyosomal associations, were kickoff described in microscopy studies in 1991. Their function remains unclear, though they were not idea to be associated with agile DNA replication, transcription, or RNA processing.[35] They have been establish to frequently associate with discrete domains defined by dumbo localization of the transcription factor PTF, which promotes transcription of pocket-sized nuclear RNA (snRNA).[36]
PML bodies
Promyelocytic leukemia bodies (PML bodies) are spherical bodies institute scattered throughout the nucleoplasm, measuring around 0.1–ane.0 µm. They are known by a number of other names, including nuclear domain 10 (ND10), Kremer bodies, and PML oncogenic domains.[37] PML bodies are named after i of their major components, the promyelocytic leukemia poly peptide (PML). They are often seen in the nucleus in association with Cajal bodies and cleavage bodies.[29] Pml-/- mice, which are unable to create PML bodies, develop normally without obvious ill effects, showing that PML bodies are not required for about essential biological processes.[38]
Splicing speckles
Speckles are subnuclear structures that are enriched in pre-messenger RNA splicing factors and are located in the interchromatin regions of the nucleoplasm of mammalian cells.[39] At the fluorescence-microscope level they announced equally irregular, punctate structures, which vary in size and shape, and when examined by electron microscopy they are seen as clusters of interchromatin granules. Speckles are dynamic structures, and both their protein and RNA-poly peptide components tin can bike continuously between speckles and other nuclear locations, including active transcription sites. Speckles can work with p53 as enhancers of gene activity to straight enhance the activity of sure genes. Moreover, speckle-associating and non-associating p53 gene targets are functionally singled-out.[xl]
Studies on the composition, structure and behaviour of speckles accept provided a model for agreement the functional compartmentalization of the nucleus and the organization of the gene-expression machinery[41] splicing snRNPs[42] [43] and other splicing proteins necessary for pre-mRNA processing.[41] Because of a jail cell's changing requirements, the composition and location of these bodies changes according to mRNA transcription and regulation via phosphorylation of specific proteins.[44] The splicing speckles are likewise known as nuclear speckles (nuclear specks), splicing cistron compartments (SF compartments), interchromatin granule clusters (IGCs), and B snurposomes.[45] B snurposomes are found in the amphibian oocyte nuclei and in Drosophila melanogaster embryos. B snurposomes appear lonely or attached to the Cajal bodies in the electron micrographs of the amphibian nuclei.[46] IGCs function as storage sites for the splicing factors.[47]
Paraspeckles
Discovered past Fox et al. in 2002, paraspeckles are irregularly shaped compartments in the interchromatin space of the nucleus.[48] First documented in HeLa cells, where at that place are generally ten–30 per nucleus,[49] paraspeckles are now known to also exist in all human being primary cells, transformed cell lines, and tissue sections.[50] Their proper noun is derived from their distribution in the nucleus; the "para" is short for parallel and the "speckles" refers to the splicing speckles to which they are always in close proximity.[49]
Paraspeckles sequester nuclear proteins and RNA and thus appear to office as a molecular sponge[51] that is involved in the regulation of factor expression.[52] Furthermore, paraspeckles are dynamic structures that are altered in response to changes in cellular metabolic action. They are transcription dependent[48] and in the absenteeism of RNA Political leader 2 transcription, the paraspeckle disappears and all of its associated protein components (PSP1, p54nrb, PSP2, CFI(m)68, and PSF) form a crescent shaped perinucleolar cap in the nucleolus. This miracle is demonstrated during the prison cell cycle. In the cell cycle, paraspeckles are present during interphase and during all of mitosis except for telophase. During telophase, when the two daughter nuclei are formed, there is no RNA Pol II transcription so the protein components instead form a perinucleolar cap.[50]
Perichromatin fibrils
Perichromatin fibrils are visible only nether electron microscope. They are located next to the transcriptionally active chromatin and are hypothesized to exist the sites of active pre-mRNA processing.[47]
Clastosomes
Clastosomes are small nuclear bodies (0.2–0.5 µm) described as having a thick ring-shape due to the peripheral capsule around these bodies.[27] This name is derived from the Greek klastos, broken and soma, trunk.[27] Clastosomes are not typically present in normal cells, making them hard to detect. They form under high proteolytic conditions inside the nucleus and dethrone once at that place is a subtract in activity or if cells are treated with proteasome inhibitors.[27] [53] The scarcity of clastosomes in cells indicates that they are non required for proteasome function.[54] Osmotic stress has too been shown to crusade the formation of clastosomes.[55] These nuclear bodies contain catalytic and regulatory subunits of the proteasome and its substrates, indicating that clastosomes are sites for degrading proteins.[54]
Function
The nucleus provides a site for genetic transcription that is segregated from the location of translation in the cytoplasm, allowing levels of gene regulation that are non bachelor to prokaryotes. The main function of the cell nucleus is to control gene expression and mediate the replication of Dna during the jail cell cycle.[7] : 171
Jail cell compartmentalization
The nuclear envelope allows the nucleus to command its contents, and split them from the rest of the cytoplasm where necessary. This is of import for controlling processes on either side of the nuclear membrane. In most cases where a cytoplasmic process needs to be restricted, a key participant is removed to the nucleus, where information technology interacts with transcription factors to downregulate the production of sure enzymes in the pathway. This regulatory mechanism occurs in the case of glycolysis, a cellular pathway for breaking down glucose to produce energy. Hexokinase is an enzyme responsible for the outset the footstep of glycolysis, forming glucose-6-phosphate from glucose. At high concentrations of fructose-6-phosphate, a molecule made afterwards from glucose-6-phosphate, a regulator protein removes hexokinase to the nucleus,[56] where information technology forms a transcriptional repressor complex with nuclear proteins to reduce the expression of genes involved in glycolysis.[57]
In gild to command which genes are beingness transcribed, the cell separates some transcription factor proteins responsible for regulating gene expression from physical access to the Dna until they are activated by other signaling pathways. This prevents fifty-fifty low levels of inappropriate gene expression. For case, in the instance of NF-κB-controlled genes, which are involved in well-nigh inflammatory responses, transcription is induced in response to a point pathway such as that initiated by the signaling molecule TNF-α, binds to a cell membrane receptor, resulting in the recruitment of signalling proteins, and somewhen activating the transcription factor NF-κB. A nuclear localisation signal on the NF-κB poly peptide allows it to be transported through the nuclear pore and into the nucleus, where it stimulates the transcription of the target genes.[ix]
The compartmentalization allows the jail cell to prevent translation of unspliced mRNA.[58] Eukaryotic mRNA contains introns that must be removed earlier being translated to produce functional proteins. The splicing is washed inside the nucleus before the mRNA can exist accessed by ribosomes for translation. Without the nucleus, ribosomes would interpret newly transcribed (unprocessed) mRNA, resulting in malformed and nonfunctional proteins.[vii] : 108–15
Replication
The main part of the cell nucleus is to control gene expression and mediate the replication of Dna during the cell wheel.[7] : 171 It has been found that replication happens in a localised style in the cell nucleus. In the S stage of interphase of the cell bicycle; replication takes place. Contrary to the traditional view of moving replication forks forth stagnant Deoxyribonucleic acid, a concept of replication factories emerged, which means replication forks are concentrated towards some immobilised 'mill' regions through which the template Dna strands pass like conveyor belts.[59]
Gene expression

A generic transcription manufacturing plant during transcription, highlighting the possibility of transcribing more than than i gene at a time. The diagram includes 8 RNA polymerases even so the number can vary depending on cell type. The image besides includes transcription factors and a porous, protein core.
Factor expression first involves transcription, in which Deoxyribonucleic acid is used as a template to produce RNA. In the instance of genes encoding proteins, that RNA produced from this procedure is messenger RNA (mRNA), which then needs to be translated by ribosomes to form a protein. Every bit ribosomes are located outside the nucleus, mRNA produced needs to be exported.[60]
Since the nucleus is the site of transcription, it also contains a multifariousness of proteins that either directly mediate transcription or are involved in regulating the process. These proteins include helicases, which unwind the double-stranded Deoxyribonucleic acid molecule to facilitate access to it, RNA polymerases, which bind to the Deoxyribonucleic acid promoter to synthesize the growing RNA molecule, topoisomerases, which change the amount of supercoiling in DNA, helping information technology current of air and unwind, likewise as a large variety of transcription factors that regulate expression.[61]
Processing of pre-mRNA
Newly synthesized mRNA molecules are known every bit primary transcripts or pre-mRNA. They must undergo mail service-transcriptional modification in the nucleus before being exported to the cytoplasm; mRNA that appears in the cytoplasm without these modifications is degraded rather than used for protein translation. The three main modifications are v' capping, 3' polyadenylation, and RNA splicing. While in the nucleus, pre-mRNA is associated with a variety of proteins in complexes known every bit heterogeneous ribonucleoprotein particles (hnRNPs). Improver of the 5' cap occurs co-transcriptionally and is the first step in postal service-transcriptional modification. The 3' poly-adenine tail is only added after transcription is complete.[7] : 509–eighteen
RNA splicing, carried out by a circuitous called the spliceosome, is the process past which introns, or regions of Deoxyribonucleic acid that do not code for poly peptide, are removed from the pre-mRNA and the remaining exons connected to re-form a unmarried continuous molecule. This procedure usually occurs afterwards v' capping and three' polyadenylation but tin can begin before synthesis is complete in transcripts with many exons.[7] : 494 Many pre-mRNAs can be spliced in multiple ways to produce different mature mRNAs that encode unlike protein sequences. This process is known as alternative splicing, and allows production of a large multifariousness of proteins from a limited corporeality of Deoxyribonucleic acid.[62]
Dynamics and regulation
Nuclear send

The entry and exit of big molecules from the nucleus is tightly controlled past the nuclear pore complexes. Although small molecules can enter the nucleus without regulation,[63] macromolecules such as RNA and proteins require association karyopherins called importins to enter the nucleus and exportins to exit. "Cargo" proteins that must be translocated from the cytoplasm to the nucleus contain short amino acid sequences known as nuclear localization signals, which are bound by importins, while those transported from the nucleus to the cytoplasm conduct nuclear export signals jump past exportins. The power of importins and exportins to transport their cargo is regulated by GTPases, enzymes that hydrolyze the molecule guanosine triphosphate (GTP) to release energy. The central GTPase in nuclear transport is Ran, which is jump to either GTP or Gross domestic product (guanosine diphosphate), depending on whether it is located in the nucleus or the cytoplasm. Whereas importins depend on RanGTP to dissociate from their cargo, exportins require RanGTP in social club to demark to their cargo.[eight]
Nuclear import depends on the importin binding its cargo in the cytoplasm and conveying it through the nuclear pore into the nucleus. Inside the nucleus, RanGTP acts to divide the cargo from the importin, allowing the importin to get out the nucleus and exist reused. Nuclear consign is like, equally the exportin binds the cargo inside the nucleus in a process facilitated by RanGTP, exits through the nuclear pore, and separates from its cargo in the cytoplasm.[64]
Specialized export proteins exist for translocation of mature mRNA and tRNA to the cytoplasm after mail-transcriptional modification is consummate. This quality-command mechanism is of import due to these molecules' fundamental role in poly peptide translation. Mis-expression of a protein due to incomplete excision of exons or mis-incorporation of amino acids could have negative consequences for the cell; thus, incompletely modified RNA that reaches the cytoplasm is degraded rather than used in translation.[seven]
Assembly and disassembly

During its lifetime, a nucleus may be cleaved down or destroyed, either in the process of jail cell division or as a issue of apoptosis (the process of programmed cell death). During these events, the structural components of the nucleus — the envelope and lamina — can be systematically degraded. In almost cells, the disassembly of the nuclear envelope marks the stop of the prophase of mitosis. Even so, this disassembly of the nucleus is non a universal characteristic of mitosis and does not occur in all cells. Some unicellular eukaryotes (eastward.chiliad., yeasts) undergo then-called closed mitosis, in which the nuclear envelope remains intact. In closed mitosis, the daughter chromosomes migrate to opposite poles of the nucleus, which then divides in ii. The cells of college eukaryotes, however, usually undergo open up mitosis, which is characterized by breakup of the nuclear envelope. The daughter chromosomes then migrate to opposite poles of the mitotic spindle, and new nuclei reassemble effectually them.[7] : 854
At a sure point during the jail cell bike in open mitosis, the cell divides to form two cells. In order for this process to exist possible, each of the new daughter cells must take a full set up of genes, a process requiring replication of the chromosomes likewise every bit segregation of the carve up sets. This occurs by the replicated chromosomes, the sister chromatids, attaching to microtubules, which in plow are attached to different centrosomes. The sister chromatids tin can and so be pulled to split locations in the cell. In many cells, the centrosome is located in the cytoplasm, outside the nucleus; the microtubules would be unable to adhere to the chromatids in the presence of the nuclear envelope.[65] Therefore, the early stages in the jail cell bike, get-go in prophase and until around prometaphase, the nuclear membrane is dismantled.[12] Likewise, during the same period, the nuclear lamina is besides disassembled, a process regulated by phosphorylation of the lamins by protein kinases such as the CDC2 protein kinase.[66] Towards the cease of the prison cell cycle, the nuclear membrane is reformed, and around the same time, the nuclear lamina are reassembled by dephosphorylating the lamins.[66]
However, in dinoflagellates, the nuclear envelope remains intact, the centrosomes are located in the cytoplasm, and the microtubules come in contact with chromosomes, whose centromeric regions are incorporated into the nuclear envelope (the so-called closed mitosis with extranuclear spindle). In many other protists (e.g., ciliates, sporozoans) and fungi, the centrosomes are intranuclear, and their nuclear envelope also does not detach during cell division.[67]
Apoptosis is a controlled process in which the prison cell's structural components are destroyed, resulting in death of the cell. Changes associated with apoptosis direct affect the nucleus and its contents, for example, in the condensation of chromatin and the disintegration of the nuclear envelope and lamina. The destruction of the lamin networks is controlled by specialized apoptotic proteases called caspases, which cleave the lamin proteins and, thus, dethrone the nucleus' structural integrity. Lamin cleavage is sometimes used every bit a laboratory indicator of caspase activity in assays for early apoptotic activity.[12] Cells that express mutant caspase-resistant lamins are deficient in nuclear changes related to apoptosis, suggesting that lamins play a role in initiating the events that atomic number 82 to apoptotic degradation of the nucleus.[12] Inhibition of lamin assembly itself is an inducer of apoptosis.[68]
The nuclear envelope acts every bit a barrier that prevents both DNA and RNA viruses from inbound the nucleus. Some viruses crave access to proteins inside the nucleus in order to replicate and/or assemble. DNA viruses, such as herpesvirus replicate and get together in the cell nucleus, and exit by budding through the inner nuclear membrane. This process is accompanied past disassembly of the lamina on the nuclear face of the inner membrane.[12]
Initially, it has been suspected that immunoglobulins in general and autoantibodies in item do non enter the nucleus. At present there is a body of prove that under pathological weather condition (eastward.1000. lupus erythematosus) IgG tin can enter the nucleus.[69]
Nuclei per cell
Most eukaryotic jail cell types usually have a single nucleus, but some have no nuclei, while others take several. This can outcome from normal development, as in the maturation of mammalian red blood cells, or from faulty jail cell sectionalization.[70]
Anucleated cells
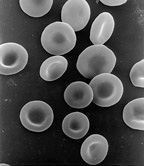
Human red blood cells, like those of other mammals, lack nuclei. This occurs equally a normal function of the cells' development.
An anucleated cell contains no nucleus and is, therefore, incapable of dividing to produce daughter cells. The best-known anucleated cell is the mammalian ruddy blood cell, or erythrocyte, which also lacks other organelles such equally mitochondria, and serves primarily equally a ship vessel to ferry oxygen from the lungs to the body's tissues. Erythrocytes mature through erythropoiesis in the bone marrow, where they lose their nuclei, organelles, and ribosomes. The nucleus is expelled during the procedure of differentiation from an erythroblast to a reticulocyte, which is the immediate forerunner of the mature erythrocyte.[71] The presence of mutagens may induce the release of some immature "micronucleated" erythrocytes into the bloodstream.[72] [73] Anucleated cells can besides arise from flawed jail cell segmentation in which one daughter lacks a nucleus and the other has two nuclei.
In flowering plants, this condition occurs in sieve tube elements.[74]
Multinucleated cells
Multinucleated cells contain multiple nuclei. Most acantharean species of protozoa[75] and some fungi in mycorrhizae[76] take naturally multinucleated cells. Other examples include the intestinal parasites in the genus Giardia, which take two nuclei per cell.[77] Ciliates take ii kinds of nuclei in a unmarried jail cell, a somatic macronucleus and a germline micronucleus.[78] In humans, skeletal musculus cells, also chosen myocytes and syncytium, become multinucleated during development; the resulting organization of nuclei most the periphery of the cells allows maximal intracellular space for myofibrils.[7] Other multinucleate cells in the human are osteoclasts a type of bone cell. Multinucleated and binucleated cells can also be abnormal in humans; for case, cells arising from the fusion of monocytes and macrophages, known every bit giant multinucleated cells, sometimes back-trail inflammation[79] and are besides implicated in tumor formation.[eighty]
A number of dinoflagellates are known to accept two nuclei. Different other multinucleated cells these nuclei contain two distinct lineages of Deoxyribonucleic acid: one from the dinoflagellate and the other from a symbiotic diatom.[81]
Evolution
Equally the major defining characteristic of the eukaryotic jail cell, the nucleus' evolutionary origin has been the subject of much speculation. Four major hypotheses take been proposed to explain the existence of the nucleus, although none have yet earned widespread support.[82] [83] [84]
The beginning model known as the "syntrophic model" proposes that a symbiotic relationship between the archaea and bacteria created the nucleus-containing eukaryotic cell. (Organisms of the Archaea and Bacteria domain have no cell nucleus.[85]) It is hypothesized that the symbiosis originated when aboriginal archaea, similar to modern methanogenic archaea, invaded and lived within bacteria similar to modern myxobacteria, somewhen forming the early nucleus. This theory is analogous to the accepted theory for the origin of eukaryotic mitochondria and chloroplasts, which are thought to have developed from a similar endosymbiotic relationship between proto-eukaryotes and aerobic leaner.[86] The archaeal origin of the nucleus is supported by observations that archaea and eukarya have similar genes for certain proteins, including histones. Observations that myxobacteria are motile, tin form multicellular complexes, and possess kinases and G proteins similar to eukarya, back up a bacterial origin for the eukaryotic cell.[87]
A second model proposes that proto-eukaryotic cells evolved from bacteria without an endosymbiotic phase. This model is based on the existence of mod Planctomycetota bacteria that possess a nuclear construction with archaic pores and other compartmentalized membrane structures.[88] A similar proposal states that a eukaryote-like cell, the chronocyte, evolved first and phagocytosed archaea and bacteria to generate the nucleus and the eukaryotic cell.[89]
The nearly controversial model, known as viral eukaryogenesis, posits that the membrane-bound nucleus, along with other eukaryotic features, originated from the infection of a prokaryote by a virus. The suggestion is based on similarities between eukaryotes and viruses such as linear DNA strands, mRNA capping, and tight binding to proteins (analogizing histones to viral envelopes). One version of the proposal suggests that the nucleus evolved in concert with phagocytosis to form an early cellular "predator".[90] Another variant proposes that eukaryotes originated from early on archaea infected by poxviruses, on the basis of observed similarity between the DNA polymerases in modern poxviruses and eukaryotes.[91] [92] It has been suggested that the unresolved question of the development of sex could exist related to the viral eukaryogenesis hypothesis.[93]
A more recent proposal, the exomembrane hypothesis, suggests that the nucleus instead originated from a single ancestral jail cell that evolved a second exterior cell membrane; the interior membrane enclosing the original cell and then became the nuclear membrane and evolved increasingly elaborate pore structures for passage of internally synthesized cellular components such equally ribosomal subunits.[94]
History


The nucleus was the commencement organelle to be discovered. What is near likely the oldest preserved cartoon dates back to the early microscopist Antonie van Leeuwenhoek (1632–1723). He observed a "lumen", the nucleus, in the cherry-red blood cells of salmon.[95] Unlike mammalian red blood cells, those of other vertebrates still contain nuclei.[96]
The nucleus was also described by Franz Bauer in 1804[97] and in more detail in 1831 by Scottish botanist Robert Brown in a talk at the Linnean Society of London. Brownish was studying orchids under the microscope when he observed an opaque area, which he called the "areola" or "nucleus", in the cells of the blossom'south outer layer.[98] He did not suggest a potential office.
In 1838, Matthias Schleiden proposed that the nucleus plays a role in generating cells, thus he introduced the proper noun "cytoblast" ("cell builder"). He believed that he had observed new cells assembling around "cytoblasts". Franz Meyen was a strong opponent of this view, having already described cells multiplying by division and believing that many cells would have no nuclei. The idea that cells can be generated de novo, by the "cytoblast" or otherwise, contradicted piece of work by Robert Remak (1852) and Rudolf Virchow (1855) who decisively propagated the new paradigm that cells are generated solely past cells (" Omnis cellula e cellula "). The function of the nucleus remained unclear.[99]
Betwixt 1877 and 1878, Oscar Hertwig published several studies on the fertilization of sea urchin eggs, showing that the nucleus of the sperm enters the oocyte and fuses with its nucleus. This was the kickoff time it was suggested that an individual develops from a (unmarried) nucleated cell. This was in contradiction to Ernst Haeckel's theory that the complete phylogeny of a species would be repeated during embryonic evolution, including generation of the starting time nucleated cell from a "monerula", a structureless mass of primordial protoplasm ("Urschleim"). Therefore, the necessity of the sperm nucleus for fertilization was discussed for quite some time. However, Hertwig confirmed his observation in other animal groups, including amphibians and molluscs. Eduard Strasburger produced the same results for plants in 1884. This paved the way to assign the nucleus an important function in heredity. In 1873, August Weismann postulated the equivalence of the maternal and paternal germ cells for heredity. The part of the nucleus equally carrier of genetic data became clear only subsequently, afterward mitosis was discovered and the Mendelian rules were rediscovered at the beginning of the 20th century; the chromosome theory of heredity was therefore developed.[99]
See also
- Nucleus (neuroanatomy)
- Nucleoid
- Nucleomorph
References
- ^ Cantwell H, Nurse P (2019). "Unravelling nuclear size command". Current Genetics. Springer. 65 (six): 1282. doi:10.1007/s00294-019-00999-3. PMC6820586. PMID 31147736.
- ^ a b c Lodish HF, Berk A, Kaiser C, Krieger M, Bretscher A, Ploegh H, et al. (2016). Molecular Prison cell Biology (Eighth ed.). New York: W.H. Freeman. ISBN978-1-4641-8339-iii.
- ^ Alberts B, Johnson A, Lewis J, Raff Grand, Roberts K, Walter P (2002). Molecular biological science of the cell (4th ed.). New York: Garland Science. p. 197. ISBN978-0-8153-4072-0.
- ^ a b c d due east f Alberts B, Johnson A, Lewis J, Morgan D, Raff M, Roberts K, Walter P (2015). Molecular Biological science of the Prison cell (6 ed.). New York: Garland Science.
- ^ Rhoades R, Pflanzer R, eds. (1996). "Ch3". Human Physiology (3rd ed.). Saunders College Publishing.
- ^ Shulga N, Mosammaparast N, Wozniak R, Goldfarb DS (May 2000). "Yeast nucleoporins involved in passive nuclear envelope permeability". Main. The Journal of Cell Biology. 149 (5): 1027–38. doi:10.1083/jcb.149.5.1027. PMC2174828. PMID 10831607.
- ^ a b c d eastward f g h i j k l m Lodish H, Berk A, Matsudaira P, Kaiser CA, Krieger M, Scott MP, Zipursky SL, Darnell J (2004). Molecular Prison cell Biology (fifth ed.). New York: WH Freeman. ISBN978-0-7167-2672-two.
- ^ a b Pemberton LF, Paschal BM (March 2005). "Mechanisms of receptor-mediated nuclear import and nuclear export". Review. Traffic. 6 (3): 187–98. doi:x.1111/j.1600-0854.2005.00270.x. PMID 15702987. S2CID 172279.
- ^ a b c Alberts B, Johnson A, Lewis J, Raff One thousand, Roberts K, Walter P, eds. (2002). "Chapter 4: Deoxyribonucleic acid and Chromosomes". Molecular Biology of the Cell (4th ed.). New York: Garland Science. pp. 191–234. ISBN978-0-8153-4072-0.
- ^ Stuurman N, Heins S, Aebi U (1998). "Nuclear lamins: their structure, assembly, and interactions". Review. Journal of Structural Biology. 122 (one–2): 42–66. doi:10.1006/jsbi.1998.3987. PMID 9724605.
- ^ Goldman AE, Moir RD, Montag-Lowy M, Stewart Thousand, Goldman RD (November 1992). "Pathway of incorporation of microinjected lamin A into the nuclear envelope". Primary. The Periodical of Cell Biology. 119 (4): 725–35. doi:10.1083/jcb.119.4.725. PMC2289687. PMID 1429833.
- ^ a b c d east Goldman RD, Gruenbaum Y, Moir RD, Shumaker DK, Spann TP (March 2002). "Nuclear lamins: building blocks of nuclear architecture". Review. Genes & Development. 16 (5): 533–47. doi:10.1101/gad.960502. PMID 11877373.
- ^ Broers JL, Ramaekers FC (2004). "Dynamics of nuclear lamina assembly and disassembly". Review. Symposia of the Society for Experimental Biological science (56): 177–92. ISBN9781134279838. PMID 15565881.
- ^ Moir RD, Yoon One thousand, Khuon S, Goldman RD (December 2000). "Nuclear lamins A and B1: dissimilar pathways of assembly during nuclear envelope germination in living cells". Primary. The Journal of Cell Biology. 151 (6): 1155–68. doi:10.1083/jcb.151.6.1155. PMC2190592. PMID 11121432.
- ^ Spann TP, Goldman AE, Wang C, Huang S, Goldman RD (February 2002). "Alteration of nuclear lamin organization inhibits RNA polymerase Two-dependent transcription". Primary. The Periodical of Cell Biology. 156 (4): 603–8. doi:x.1083/jcb.200112047. PMC2174089. PMID 11854306.
- ^ Mounkes LC, Stewart CL (June 2004). "Crumbling and nuclear organisation: lamins and progeria". Review. Electric current Opinion in Cell Biology. xvi (3): 322–7. doi:ten.1016/j.ceb.2004.03.009. PMID 15145358.
- ^ Ehrenhofer-Murray AE (June 2004). "Chromatin dynamics at DNA replication, transcription and repair". Review. European Journal of Biochemistry. 271 (12): 2335–49. doi:x.1111/j.1432-1033.2004.04162.x. PMID 15182349.
- ^ Grigoryev SA, Bulynko YA, Popova EY (2006). "The end adjusts the ways: heterochromatin remodelling during terminal jail cell differentiation". Review. Chromosome Research. fourteen (1): 53–69. doi:10.1007/s10577-005-1021-6. PMID 16506096. S2CID 6040822.
- ^ Schardin Yard, Cremer T, Hager HD, Lang M (December 1985). "Specific staining of human chromosomes in Chinese hamster x man hybrid cell lines demonstrates interphase chromosome territories" (PDF). Primary. Homo Genetics. 71 (4): 281–7. doi:10.1007/BF00388452. PMID 2416668. S2CID 9261461.
- ^ Lamond AI, Earnshaw WC (April 1998). "Structure and function in the nucleus" (PDF). Review. Science. 280 (5363): 547–53. CiteSeerX10.1.i.323.5543. doi:10.1126/scientific discipline.280.5363.547. PMID 9554838.
- ^ Kurz A, Lampel South, Nickolenko JE, Bradl J, Benner A, Zirbel RM, et al. (December 1996). "Active and inactive genes localize preferentially in the periphery of chromosome territories". Primary. The Periodical of Cell Biology. 135 (v): 1195–205. doi:10.1083/jcb.135.five.1195. PMC2121085. PMID 8947544. Archived from the original on 29 September 2007.
- ^ Rothfield NF, Stollar BD (Nov 1967). "The relation of immunoglobulin grade, design of anti-nuclear antibody, and complement-fixing antibodies to Dna in sera from patients with systemic lupus erythematosus". Primary. The Journal of Clinical Investigation. 46 (11): 1785–94. doi:10.1172/JCI105669. PMC292929. PMID 4168731.
- ^ Barned S, Goodman AD, Mattson DH (Feb 1995). "Frequency of anti-nuclear antibodies in multiple sclerosis". Primary. Neurology. 45 (2): 384–5. doi:ten.1212/WNL.45.2.384. PMID 7854544. S2CID 30482028.
- ^ Hernandez-Verdun D (Jan 2006). "Nucleolus: from structure to dynamics". Review. Histochemistry and Cell Biology. 125 (one–2): 127–37. doi:ten.1007/s00418-005-0046-iv. PMID 16328431. S2CID 20769260.
- ^ a b Lamond AI, Sleeman JE (Oct 2003). "Nuclear substructure and dynamics". Review. Current Biology. 13 (21): R825-viii. doi:10.1016/j.cub.2003.10.012. PMID 14588256. S2CID 16865665.
- ^ a b c Cioce M, Lamond AI (2005). "Cajal bodies: a long history of discovery". Review. Annual Review of Cell and Developmental Biology. 21: 105–31. doi:10.1146/annurev.cellbio.twenty.010403.103738. PMID 16212489. S2CID 8807316.
- ^ a b c d Lafarga M, Berciano MT, Pena E, Mayo I, Castaño JG, Bohmann D, et al. (August 2002). "Clastosome: a subtype of nuclear body enriched in 19S and 20S proteasomes, ubiquitin, and protein substrates of proteasome". Primary. Molecular Biology of the Cell. 13 (eight): 2771–82. CiteSeerX10.one.1.321.6138. doi:10.1091/mbc.e02-03-0122. PMC117941. PMID 12181345.
- ^ a b c Pollard TD, Earnshaw WC (2004). Cell Biology . Philadelphia: Saunders. ISBN978-0-7216-3360-ii.
- ^ a b c Dundr Thou, Misteli T (June 2001). "Functional compages in the prison cell nucleus". Review. The Biochemical Journal. 356 (Pt 2): 297–310. doi:x.1042/0264-6021:3560297. PMC1221839. PMID 11368755.
- ^ Bond CS, Play a joke on AH (September 2009). "Paraspeckles: nuclear bodies congenital on long noncoding RNA". Review. The Journal of Cell Biology. 186 (5): 637–44. doi:10.1083/jcb.200906113. PMC2742191. PMID 19720872.
- ^ Goebel HH, Warlo I (January 1997). "Nemaline myopathy with intranuclear rods--intranuclear rod myopathy". Review. Neuromuscular Disorders. 7 (ane): 13–9. doi:ten.1016/S0960-8966(96)00404-X. PMID 9132135. S2CID 29584217.
- ^ a b c Matera AG, Frey MR (Baronial 1998). "Coiled bodies and gems: Janus or gemini?". Review. American Journal of Human Genetics. 63 (2): 317–21. doi:x.1086/301992. PMC1377332. PMID 9683623.
- ^ Matera AG (Baronial 1998). "Of coiled bodies, gems, and salmon". Review. Journal of Cellular Biochemistry. 70 (2): 181–92. doi:10.1002/(sici)1097-4644(19980801)70:ii<181::aid-jcb4>3.0.co;ii-thou. PMID 9671224.
- ^ Saunders WS, Cooke CA, Earnshaw WC (November 1991). "Compartmentalization within the nucleus: discovery of a novel subnuclear region". Master. The Journal of Jail cell Biology. 115 (4): 919–31. doi:10.1083/jcb.115.4.919. PMC2289954. PMID 1955462.
- ^ Pombo A, Cuello P, Schul Westward, Yoon JB, Roeder RG, Cook PR, Spud S (March 1998). "Regional and temporal specialization in the nucleus: a transcriptionally-agile nuclear domain rich in PTF, Oct1 and PIKA antigens associates with specific chromosomes early in the jail cell bicycle". Primary. The EMBO Journal. 17 (6): 1768–78. doi:10.1093/emboj/17.6.1768. PMC1170524. PMID 9501098.
- ^ Zimber A, Nguyen QD, Gespach C (October 2004). "Nuclear bodies and compartments: functional roles and cellular signalling in health and illness". Review. Cellular Signalling. 16 (10): 1085–104. doi:10.1016/j.cellsig.2004.03.020. PMID 15240004.
- ^ Lallemand-Breitenbach Five, de Thé H (May 2010). "PML nuclear bodies". Review. Common cold Bound Harbor Perspectives in Biological science. 2 (5): a000661. doi:10.1101/cshperspect.a000661. PMC2857171. PMID 20452955.
- ^ Spector DL, Lamond AI (February 2011). "Nuclear speckles". Review. Cold Spring Harbor Perspectives in Biology. iii (2): a000646. doi:ten.1101/cshperspect.a000646. PMC3039535. PMID 20926517.
- ^ Alexander KA, Coté A, Nguyen SC, Zhang L, Berger SL (March 2021). "p53 mediates target gene clan with nuclear speckles for amplified RNA expression". Primary. Molecular Cell. 81 (8): S1097-2765(21)00174-X. doi:10.1016/j.molcel.2021.03.006. PMID 33823140. S2CID 233172170.
- ^ a b Lamond AI, Spector DL (August 2003). "Nuclear speckles: a model for nuclear organelles". Review. Nature Reviews. Molecular Prison cell Biological science. 4 (8): 605–12. doi:10.1038/nrm1172. PMID 12923522. S2CID 6439413.
- ^ Tripathi K, Parnaik VK (September 2008). "Differential dynamics of splicing factor SC35 during the prison cell cycle" (PDF). Primary. Journal of Biosciences. 33 (3): 345–54. doi:ten.1007/s12038-008-0054-iii. PMID 19005234. S2CID 6332495. Archived (PDF) from the original on xv November 2011.
- ^ Tripathi K, Parnaik VK (September 2008). "Differential dynamics of splicing factor SC35 during the cell bicycle". Primary. Journal of Biosciences. 33 (3): 345–54. doi:x.1007/s12038-008-0054-3. PMID 19005234. S2CID 6332495.
- ^ Handwerger KE, Gall JG (January 2006). "Subnuclear organelles: new insights into form and function". Review. Trends in Cell Biology. sixteen (ane): 19–26. doi:10.1016/j.tcb.2005.11.005. PMID 16325406.
- ^ "Cellular component Nucleus speckle". UniProt: UniProtKB. Retrieved 30 August 2013.
- ^ Gall JG, Bellini M, Wu Z, Murphy C (December 1999). "Associates of the nuclear transcription and processing machinery: Cajal bodies (coiled bodies) and transcriptosomes". Master. Molecular Biology of the Cell. ten (12): 4385–402. doi:10.1091/mbc.x.12.4385. PMC25765. PMID 10588665.
- ^ a b Matera AG, Terns RM, Terns MP (March 2007). "Non-coding RNAs: lessons from the small nuclear and small-scale nucleolar RNAs". Review. Nature Reviews. Molecular Cell Biology. 8 (3): 209–twenty. doi:x.1038/nrm2124. PMID 17318225. S2CID 30268055.
- ^ a b Fox AH, Lamond AI (July 2010). "Paraspeckles". Review. Cold Jump Harbor Perspectives in Biology. 2 (vii): a000687. doi:10.1101/cshperspect.a000687. PMC2890200. PMID 20573717.
- ^ a b Play a joke on A, Bickmore W (2004). "Nuclear Compartments: Paraspeckles". Nuclear Protein Database. Archived from the original on 10 September 2008. Retrieved 6 March 2007.
- ^ a b Trick AH, Bail CS, Lamond AI (Nov 2005). "P54nrb forms a heterodimer with PSP1 that localizes to paraspeckles in an RNA-dependent style". Principal. Molecular Biological science of the Jail cell. xvi (11): 5304–xv. doi:10.1091/mbc.E05-06-0587. PMC1266428. PMID 16148043.
- ^ Nakagawa Due south, Yamazaki T, Hirose T (October 2018). "Molecular autopsy of nuclear paraspeckles: towards agreement the emerging world of the RNP milieu". Review. Open Biology. 8 (ten): 180150. doi:x.1098/rsob.180150. PMC6223218. PMID 30355755.
- ^ Pisani G, Businesswoman B (Dec 2019). "Nuclear paraspeckles function in mediating gene regulatory and apoptotic pathways". Review. Non-Coding RNA Research. four (four): 128–134. doi:10.1016/j.ncrna.2019.11.002. PMC7012776. PMID 32072080.
- ^ Kong XN, Yan HX, Chen Fifty, Dong LW, Yang W, Liu Q, et al. (October 2007). "LPS-induced down-regulation of bespeak regulatory protein {alpha} contributes to innate allowed activation in macrophages". Primary. The Journal of Experimental Medicine. 204 (11): 2719–31. doi:10.1084/jem.20062611. PMC2118489. PMID 17954568.
- ^ a b Carmo-Fonseca M, Berciano MT, Lafarga M (September 2010). "Orphan nuclear bodies". Review. Cold Jump Harbor Perspectives in Biological science. ii (9): a000703. doi:10.1101/cshperspect.a000703. PMC2926751. PMID 20610547.
- ^ Sampuda KM, Riley M, Boyd L (April 2017). "Stress induced nuclear granules form in response to accumulation of misfolded proteins in Caenorhabditis elegans". Primary. BMC Cell Biology. 18 (1): xviii. doi:10.1186/s12860-017-0136-x. PMC5395811. PMID 28424053.
- ^ Lehninger AL, Nelson DL, Cox MM (2000). Lehninger principles of biochemistry (tertiary ed.). New York: Worth Publishers. ISBN978-i-57259-931-4.
- ^ Moreno F, Ahuatzi D, Riera A, Palomino CA, Herrero P (February 2005). "Glucose sensing through the Hxk2-dependent signalling pathway". Master. Biochemical Gild Transactions. 33 (Pt 1): 265–8. doi:x.1042/BST0330265. PMID 15667322. S2CID 20647022.
- ^ Görlich D, Kutay U (1999). "Send between the cell nucleus and the cytoplasm". Review. Almanac Review of Jail cell and Developmental Biology. 15 (1): 607–threescore. doi:10.1146/annurev.cellbio.15.1.607. PMID 10611974.
- ^ Hozák P, Cook PR (February 1994). "Replication factories". Review. Trends in Cell Biology. 4 (ii): 48–52. doi:10.1016/0962-8924(94)90009-4. PMID 14731866.
- ^ Nierhaus KH, Wilson DN (2004). Protein Synthesis and Ribosome Structure: Translating the Genome. Wiley-VCH. ISBN978-3-527-30638-1.
- ^ Nicolini CA (1997). Genome Construction and Office: From Chromosomes Characterization to Genes Technology. Springer. ISBN978-0-7923-4565-seven.
- ^ Black DL (2003). "Mechanisms of alternative pre-messenger RNA splicing" (PDF). Review. Annual Review of Biochemistry. 72 (i): 291–336. doi:10.1146/annurev.biochem.72.121801.161720. PMID 12626338.
- ^ Watson JD, Baker TA, Bell SP, Gann A, Levine M, Losick R (2004). "Ch9–10". Molecular Biology of the Gene (5th ed.). Peason Benjamin Cummings; CSHL Press. ISBN978-0-8053-9603-4.
- ^ Cavazza T, Vernos I (2015). "The RanGTP Pathway: From Nucleo-Cytoplasmic Transport to Spindle Assembly and Beyond". Review. Frontiers in Cell and Developmental Biology. 3: 82. doi:ten.3389/fcell.2015.00082. PMC4707252. PMID 26793706.
- ^ Lippincott-Schwartz J (March 2002). "Cell biology: ripping upwardly the nuclear envelope". Commentary. Nature. 416 (6876): 31–2. Bibcode:2002Natur.416...31L. doi:10.1038/416031a. PMID 11882878. S2CID 4431000.
- ^ a b Boulikas T (1995). "Phosphorylation of transcription factors and control of the cell cycle". Review. Critical Reviews in Eukaryotic Gene Expression. v (1): 1–77. PMID 7549180.
- ^ Boettcher B, Barral Y (2013). "The cell biology of open up and closed mitosis". Review. Nucleus. Austin, Tex. 4 (iii): 160–5. doi:10.4161/nucl.24676. PMC3720745. PMID 23644379.
- ^ Steen RL, Collas P (Apr 2001). "Mistargeting of B-blazon lamins at the stop of mitosis: implications on prison cell survival and regulation of lamins A/C expression". Master. The Journal of Cell Biological science. 153 (iii): 621–six. doi:10.1083/jcb.153.three.621. PMC2190567. PMID 11331311.
- ^ Böhm I (November 2007). "IgG deposits tin can be detected in cell nuclei of patients with both lupus erythematosus and malignancy". Primary. Clinical Rheumatology. 26 (eleven): 1877–82. doi:x.1007/s10067-007-0597-y. PMID 17364135. S2CID 44879431.
- ^ Ressel L (2017). "Nuclear Morphologies". Normal cell morphology in canine and feline cytology: an identification guide. Hoboken, NJ: John Wiley & Sons. p. 6. ISBN978-i-119-27891-7.
- ^ Skutelsky Due east, Danon D (June 1970). "Comparative written report of nuclear expulsion from the late erythroblast and cytokinesis". Principal. Experimental Cell Inquiry. 60 (three): 427–36. doi:10.1016/0014-4827(70)90536-seven. PMID 5422968.
- ^ Torous DK, Dertinger SD, Hall NE, Tometsko CR (February 2000). "Enumeration of micronucleated reticulocytes in rat peripheral blood: a flow cytometric study". Primary. Mutation Enquiry. 465 (1–2): 91–9. doi:10.1016/S1383-5718(99)00216-eight. PMID 10708974.
- ^ Hutter KJ, Stöhr M (1982). "Rapid detection of mutagen induced micronucleated erythrocytes by catamenia cytometry". Primary. Histochemistry. 75 (3): 353–62. doi:10.1007/bf00496738. PMID 7141888. S2CID 28973947.
- ^ Ham BK, Lucas WJ (April 2014). "The angiosperm phloem sieve tube arrangement: a office in mediating traits important to modern agronomics". Journal of Experimental Botany. 65 (7): 1799–816. doi:ten.1093/jxb/ert417. PMID 24368503.
- ^ Zettler LA, Sogin ML, Caron DA (October 1997). "Phylogenetic relationships betwixt the Acantharea and the Polycystinea: a molecular perspective on Haeckel'southward Radiolaria". Primary. Proceedings of the National Academy of Sciences of the U.s. of America. 94 (21): 11411–half dozen. Bibcode:1997PNAS...9411411A. doi:10.1073/pnas.94.21.11411. PMC23483. PMID 9326623.
- ^ Horton TR (2006). "The number of nuclei in basidiospores of 63 species of ectomycorrhizal Homobasidiomycetes". Primary. Mycologia. 98 (2): 233–viii. doi:10.3852/mycologia.98.two.233. PMID 16894968.
- ^ Adam RD (December 1991). "The biology of Giardia spp". Review. Microbiological Reviews. 55 (4): 706–32. doi:x.1128/MMBR.55.iv.706-732.1991. PMC372844. PMID 1779932.
- ^ Vogt A, Goldman AD, Mochizuki K, Landweber LF (1 August 2013). "Transposon Domestication versus Mutualism in Ciliate Genome Rearrangements". PLOS Genetics. 9 (viii): e1003659. doi:10.1371/journal.pgen.1003659. PMC3731211. PMID 23935529.
- ^ McInnes A, Rennick DM (February 1988). "Interleukin four induces cultured monocytes/macrophages to grade giant multinucleated cells". Chief. The Journal of Experimental Medicine. 167 (2): 598–611. doi:ten.1084/jem.167.2.598. PMC2188835. PMID 3258008.
- ^ Goldring SR, Roelke MS, Petrison KK, Bhan AK (February 1987). "Human giant cell tumors of os identification and label of cell types". Master. The Journal of Clinical Investigation. 79 (ii): 483–91. doi:10.1172/JCI112838. PMC424109. PMID 3027126.
- ^ Imanian B, Pombert JF, Dorrell RG, Burki F, Keeling PJ (2012). "Tertiary endosymbiosis in two dinotoms has generated little change in the mitochondrial genomes of their dinoflagellate hosts and diatom endosymbionts". Principal. PLOS Ane. 7 (viii): e43763. Bibcode:2012PLoSO...743763I. doi:10.1371/journal.pone.0043763. PMC3423374. PMID 22916303.
- ^ Pennisi Due east (August 2004). "Evolutionary biology. The nascence of the nucleus". News. Science. 305 (5685): 766–8. doi:10.1126/scientific discipline.305.5685.766. PMID 15297641. S2CID 83769250.
- ^ Devos DP, Gräf R, Field MC (June 2014). "Evolution of the nucleus". Review. Current Stance in Cell Biology. 28: 8–15. doi:x.1016/j.ceb.2014.01.004. PMC4071446. PMID 24508984.
- ^ López-García P, Moreira D (November 2015). "Open Questions on the Origin of Eukaryotes". Review. Trends in Environmental & Evolution. 30 (xi): 697–708. doi:10.1016/j.tree.2015.09.005. PMC4640172. PMID 26455774.
- ^ Hogan CM (2010). "Archaea". In Monosson Eastward, Cleveland C (eds.). Encyclopedia of Globe. Washington, DC.: National Council for Science and the Environment. Archived from the original on 11 May 2011.
- ^ Margulis L (1981). Symbiosis in Prison cell Evolution. San Francisco: W. H. Freeman and Company. pp. 206–227. ISBN978-0-7167-1256-5.
- ^ López-García P, Moreira D (May 2006). "Selective forces for the origin of the eukaryotic nucleus". Review. BioEssays. 28 (five): 525–33. doi:10.1002/bies.20413. PMID 16615090.
- ^ Fuerst JA (2005). "Intracellular compartmentation in planctomycetes". Review. Almanac Review of Microbiology. 59: 299–328. doi:10.1146/annurev.micro.59.030804.121258. PMID 15910279.
- ^ Hartman H, Fedorov A (Feb 2002). "The origin of the eukaryotic jail cell: a genomic investigation". Chief. Proceedings of the National Academy of Sciences of the United states of america of America. 99 (3): 1420–5. Bibcode:2002PNAS...99.1420H. doi:10.1073/pnas.032658599. PMC122206. PMID 11805300.
- ^ Bell PJ (September 2001). "Viral eukaryogenesis: was the ancestor of the nucleus a complex Dna virus?". Comment. Journal of Molecular Evolution. 53 (iii): 251–6. Bibcode:2001JMolE..53..251L. doi:ten.1007/s002390010215. PMID 11523012. S2CID 20542871.
- ^ Takemura M (May 2001). "Poxviruses and the origin of the eukaryotic nucleus". Primary. Journal of Molecular Evolution. 52 (5): 419–25. Bibcode:2001JMolE..52..419T. doi:ten.1007/s002390010171. PMID 11443345. S2CID 21200827.
- ^ Villarreal LP, DeFilippis VR (August 2000). "A hypothesis for DNA viruses as the origin of eukaryotic replication proteins". Primary. Journal of Virology. 74 (15): 7079–84. doi:10.1128/JVI.74.15.7079-7084.2000. PMC112226. PMID 10888648.
- ^ Bell PJ (November 2006). "Sex and the eukaryotic cell cycle is consistent with a viral ancestry for the eukaryotic nucleus". Primary. Periodical of Theoretical Biology. 243 (1): 54–63. Bibcode:2006JThBi.243...54B. doi:10.1016/j.jtbi.2006.05.015. PMID 16846615.
- ^ de Roos Advertisement (2006). "The origin of the eukaryotic cell based on conservation of existing interfaces". Master. Artificial Life. 12 (iv): 513–23. doi:10.1162/artl.2006.12.4.513. PMID 16953783. S2CID 5963228.
- ^ Van Leeuwenhoek A. Opera Omnia, seu Arcana Naturae ope exactissimorum Microscopiorum detecta, experimentis variis comprobata, Epistolis advertising varios illustres viros J. Arnold et Delphis, A. Beman, Lugdinum Batavorum [The Works of, or arcana of nature by means of exactissimorum microscopes had been detected and confirmed by a variety of experiments, the Epistles to the various illustrious men of valor J. Arnold and Delphi, A. Beman, Lugdina York 1719-1730] (in Latin). Cited in Gerlach D (2009). Geschichte der Mikroskopie. Frankfurt am Chief, Germany: Verlag Harri Deutsch. ISBN978-3-8171-1781-9.
- ^ Cohen WD (1982). "The cytomorphic system of anucleate non-mammalian erythrocytes". Protoplasma. 113: 23–32. doi:10.1007/BF01283036. S2CID 41287948.
- ^ Harris H (1999). The Birth of the Cell . New Haven: Yale Academy Press. ISBN978-0-300-07384-3.
- ^ Brownish R (1866). "On the Organs and Mode of Fecundation of Orchidex and Asclepiadea". Miscellaneous Botanical Works I: 511–514.
- ^ a b Cremer T (1985). Von der Zellenlehre zur Chromosomentheorie. Berlin, Heidelberg, New York, Tokyo: Springer Verlag. ISBN978-3-540-13987-4. Online Version here
Further reading
- Goldman RD, Gruenbaum Y, Moir RD, Shumaker DK, Spann TP (March 2002). "Nuclear lamins: building blocks of nuclear architecture". Genes & Development. 16 (5): 533–47. doi:ten.1101/gad.960502. PMID 11877373.
- A review article about nuclear lamins, explaining their structure and various roles
- Görlich D, Kutay U (1999). "Transport between the jail cell nucleus and the cytoplasm". Annual Review of Cell and Developmental Biological science. 15: 607–threescore. doi:10.1146/annurev.cellbio.fifteen.one.607. PMID 10611974.
- A review article virtually nuclear transport, explains the principles of the mechanism, and the various transport pathways
- Lamond AI, Earnshaw WC (April 1998). "Structure and function in the nucleus" (PDF). Science. 280 (5363): 547–53. CiteSeerX10.ane.one.323.5543. doi:x.1126/scientific discipline.280.5363.547. PMID 9554838.
- A review article about the nucleus, explaining the structure of chromosomes within the organelle, and describing the nucleolus and other subnuclear bodies
- Pennisi Due east (Baronial 2004). "Evolutionary biology. The birth of the nucleus". Science. 305 (5685): 766–8. doi:10.1126/science.305.5685.766. PMID 15297641. S2CID 83769250.
- A review commodity about the evolution of the nucleus, explaining a number of dissimilar theories
- Pollard TD, Earnshaw WC (2004). Cell Biology. Philadelphia: Saunders. ISBN978-0-7216-3360-2.
- A university level textbook focusing on cell biology. Contains data on nucleus structure and function, including nuclear send, and subnuclear domains
External links
- "The Nucleus". MBInfo.
- "Learn about the Cell Nucleus". cellnucleus.com. Website covering construction and role of the nucleus from the Department of Oncology at the University of Alberta.
- Bickmore Due west. "The Nuclear Protein Database". Medical Enquiry Council Homo Genetics Unit of measurement. Data on nuclear components.
- "The Nucleus Drove". Prototype & Video Library. The American Lodge for Jail cell Biology. Archived from the original on 12 Nov 2006. contains peer-reviewed still images and video clips that illustrate the nucleus.
- Gall JG, McIntosh JR (eds.). "Nuclear Envelope and Nuclear Import Section". Landmark Papers in Prison cell Biology. Archived from the original on 17 November 2006. contains digitized commentaries and links to seminal research papers on the nucleus. Published online in the Image & Video Library Archived 10 June 2011 at the Wayback Automobile of The American Order for Cell Biology
- "Cytoplasmic patterns generated by man antibodies". AntibodyPatterns.com. Archived from the original on 2 Jan 2007.
Source: https://en.wikipedia.org/wiki/Cell_nucleus
Posted by: florencesontoort.blogspot.com

0 Response to "(02.03 Mc) How Does The Location Of Dna In Animal Cells Compare To Dna's Location In Plant Cells?"
Post a Comment